The rationale for this effort is that left main size tends to be more stable compared with all other vessels, and a "magic number" could achieve better reproducibility. 7 The ability of providing automated volumetric lumen segmentation is a potential differential of OCT versus current IVUS systems.ĭedicated investigation, mostly on IVUS literature, has been conducted to characterize the significance of left main disease based on anatomical features and lumen geometry. 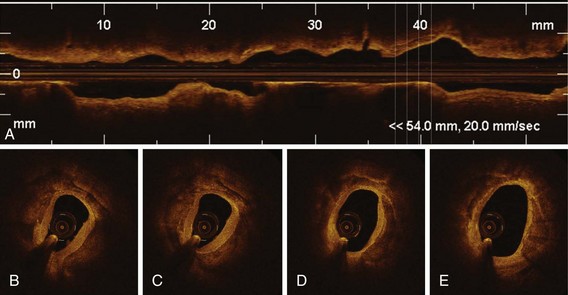
Full volumetric lumen analysis by OCT outperformed single cross-section analysis. In addition, the higher resolution of the method allows for a more automated software segmentation of the lumen with less human input, leading to a higher reproducibility. 6 This again could be explained by the higher contrast between lumen and vessel wall because blood needs to be displaced with intravascular OCT. Reference vessel lumen analyses as well as perfect circular phantom models show excellent agreement between intravascular OCT and IVUS, with lower interobserver variability obtained with intravascular OCT. 5 We speculate that the higher resolution and contrast between lumen and vessel wall obtained with intravascular OCT allow for a more detailed lumen segmentation compared with IVUS, which is mostly pronounced in irregular calcified plaque segments. Intravascular OCT-derived minimal lumen area (MLA) has a tendency to be smaller when compared with IVUS. 3,4 The large spectrum of vessel sizes and microvascular recruitment precludes single cross-section lumen analysis to accurately assess the physiological significance of a lesion. Like intravascular ultrasound (IVUS), intravascular OCT is unable to accurately predict physiology when compared with today's gold standard, fractional flow reserve (FFR). Other potentially significant findings revealed by intravascular OCT after stent implantation are stent malapposition, 1 edge dissections, 2 and tissue prolapse. The following are common questions about lesion preparation: Is there a need for atherectomy? Is there a need for thrombectomy? If pre-dilatation is chosen over direct stenting, how aggressive should the procedure be (i.e., type of balloon, balloon size, and pressure based on luminal references)? As for stent selection, what length (i.e., selecting the appropriate landing zone by avoiding landing stent in soft or calcified plaques to minimize potential edge dissections) and diameter are appropriate? Should you choose multiple stents or a single stent? If a need exists for overlapping stents, where is the best location? When it comes to stent optimization, the most studied metric related to outcomes post stent is stent expansion. For both populations, the fundamental questions are the following: Is there a merit for intervention? If so, which vessel/lesion? In terms of procedure planning, intravascular OCT can help with planning every coronary intervention procedure. In diagnostic terms, it is essential to separate two very distinct populations: Stable coronary artery disease (CAD) and acute coronary syndromes (ACS). Intravascular optical coherence tomography (OCT) can add value to angiography as a diagnostic and/or intervention tool for percutaneous coronary intervention (PCI) guidance. Editor's Note: This is Part I of a two-part Expert Analysis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
